
Certification as Minimum Threshold: Why Quality Systems Establish Acceptability, Not Differentiation
Structural Gaps in Product Information Systems — Governance & Quality
Synopsis
Certification systems serve an important institutional role: they establish whether a product meets a defined minimum standard for market entry. This paper explains why that bounded function — admissibility — should not be conflated with intelligibility, and why policy and disclosure infrastructure need separate layers to communicate producer-level differentiation across jurisdictions.
Certification establishes a minimum threshold; it does not explain what distinguishes one producer from another. This paper diagnoses the structural gap and proposes information-layer approaches.
Keywords
Certification as Minimum Threshold: Why Quality Systems Establish Acceptability, Not Differentiation
PAPER
STRUCTURAL GAPS IN PRODUCT INFORMATION SYSTEMS SGPIS-GQ — Governance & Quality
Certification as Minimum Threshold
Why Quality Systems Establish Acceptability, Not Differentiation
Paper SGPIS-GQ-01 Altibbe Inc. | March 2026
EXECUTIVE SUMMARY
Food certification systems perform a specific and valuable institutional function: they establish whether a product meets a defined minimum standard for entry into a scheme or market. They do not perform a second function that cross-border food trade increasingly requires — communicating what distinguishes one product from another within that threshold.
This paper argues that certification is an admissibility instrument, not an intelligibility instrument. Its structural limits are not a product of poor design. They arise from the gap between the bounded informational function certification was designed to perform and the broader informational role that global food trade now asks it to carry.
Evidence drawn from the European Union, the United States, Japan, India, and the Codex Alimentarius framework reveals persistent cross-jurisdictional incompatibility, asymmetric documentation burden, and a structural gap between what certification captures — conformity to scheme criteria — and what institutional buyers, trade agencies, and regulators increasingly need: producer-level differentiation, practice detail, and cross-system legibility.
The structural consequence is significant: as certification became the dominant informational signal in premium food trade, it progressively displaced richer producer-level documentation that it was never designed to replace. That gap is not addressable at the layer of certification architecture. It is a structural feature of how threshold instruments work — and it requires informational infrastructure of a different kind, one that operates above the threshold, not instead of it.
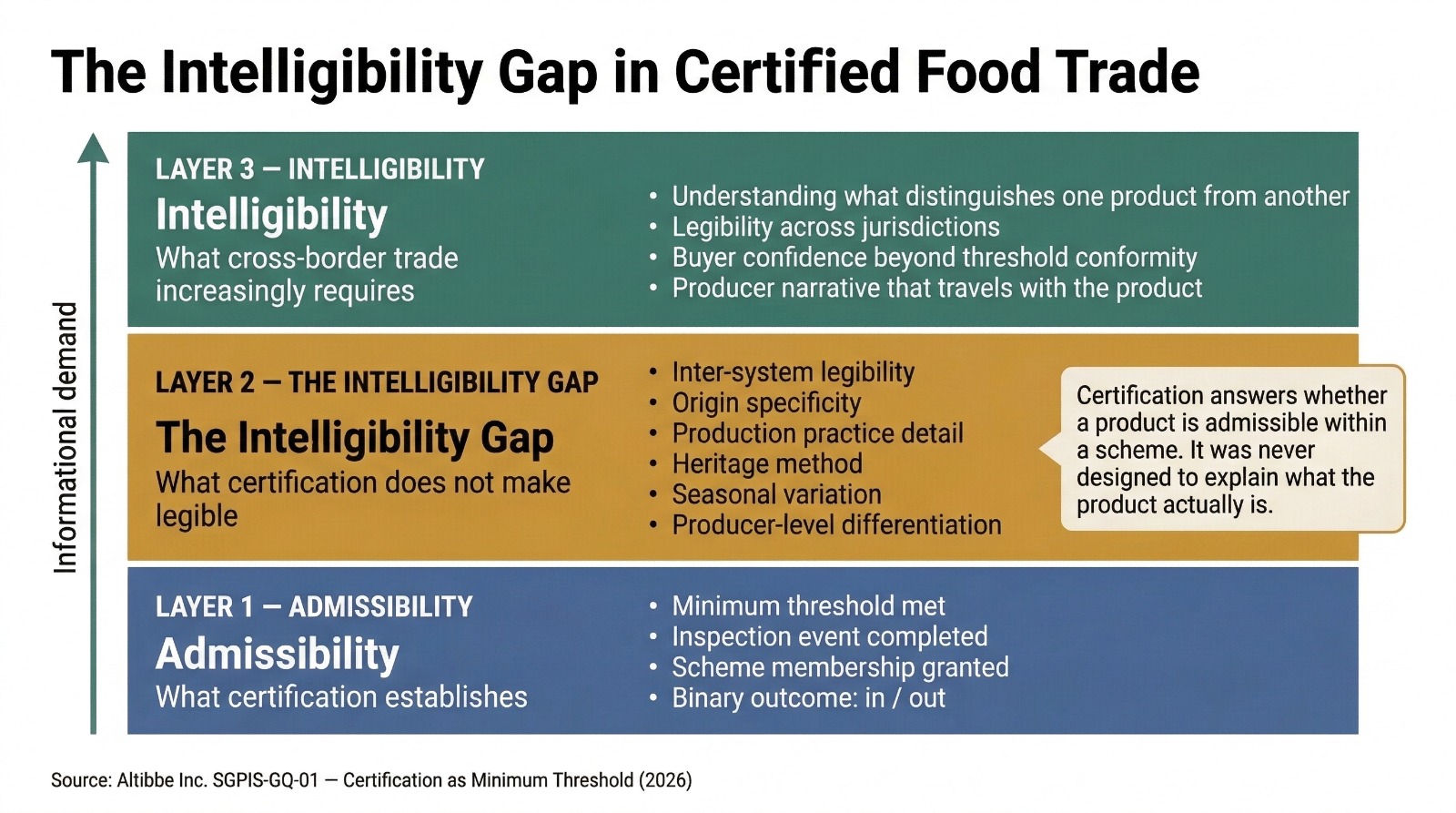
"Certification answers whether a product is admissible within a scheme. It was never designed to explain what the product actually is."
This distinction — admissibility versus intelligibility — is the paper's central structural observation.
Admissibility is what certification establishes: a product has met a defined minimum threshold, undergone a prescribed inspection or audit process, and been granted scheme membership. The threshold is specific, binary, and programme-defined.
Intelligibility is what cross-border trade increasingly requires: the ability to understand what distinguishes one product from another in terms of origin specificity, production method, heritage practice, seasonal variation, and qualitative characteristic — beyond the shared fact that both have cleared the same minimum bar.
Certification was designed to produce admissibility. The structural problem arises from the progressive displacement of the second by the first.
Three features of certification architecture drive this displacement:
Threshold compression. Certification schemes are, by design, threshold instruments. They define a minimum acceptable set of practices and award scheme membership to producers who meet that minimum. A farm that has maintained organic practices for thirty years, documents seasonal variation, and employs minimal-intervention heritage methods receives the same certification mark as a farm that converted last year and meets the letter of permitted input requirements. Both marks are honest. Neither communicates differentiation. The scheme was designed to establish acceptability, and it does — at the cost of collapsing the informational distance between producers who are very different in everything except threshold conformity.
Cross-jurisdictional fragmentation. National and regional certification systems were developed largely independently, calibrated to domestic regulatory contexts, and do not automatically translate across borders. A product certified as organic under one major system is not automatically recognised as organic under another. The implications for exporters serving multiple destination markets are significant: the same underlying practices must be demonstrated separately to each destination system, through a distinct documentation process, to a distinct set of inspectors, with a distinct permitted substances list as the standard. Multi-system certification creates documentation burden across systems that share the same substantive aspiration but do not share a common architecture.
Documentation redundancy. For exporters maintaining certification across multiple systems simultaneously, the administrative load grows with the number of destination markets — not because the underlying practices change, but because the proof architecture of each system is independent of the others. This asymmetry falls most heavily on producers with the fewest resources to absorb it.
2A. Why This Is Not a Design Failure
The limitations described above are not the result of poor institutional design or regulatory negligence. Certification systems were built to answer specific institutional questions with instruments calibrated to those questions. They have, broadly, done so effectively.
The strongest case for the current architecture is coherent: certification's value derives precisely from its specificity. A scheme that attempts to capture everything — differentiation, heritage, nutritional context, and production narrative alongside minimum practice compliance — risks losing the definitional clarity that makes baseline assurance legible, enforceable, and legally defensible. Scheme integrity requires bounded scope. A certification that means everything means nothing. Regulators and scheme administrators who designed these systems made a reasonable choice when they defined their scope narrowly.
3. Evidence and Examples
Organic Certification: Cross-System Architecture
The four major organic certification systems most relevant to global food trade — EU Regulation 2018/848, the USDA National Organic Program (NOP), the Japan Agricultural Standard (JAS) organic system, and India's National Programme for Organic Production (NPOP) — share a common aspiration: to identify products produced in conformity with defined organic principles. They do not share a common architecture.
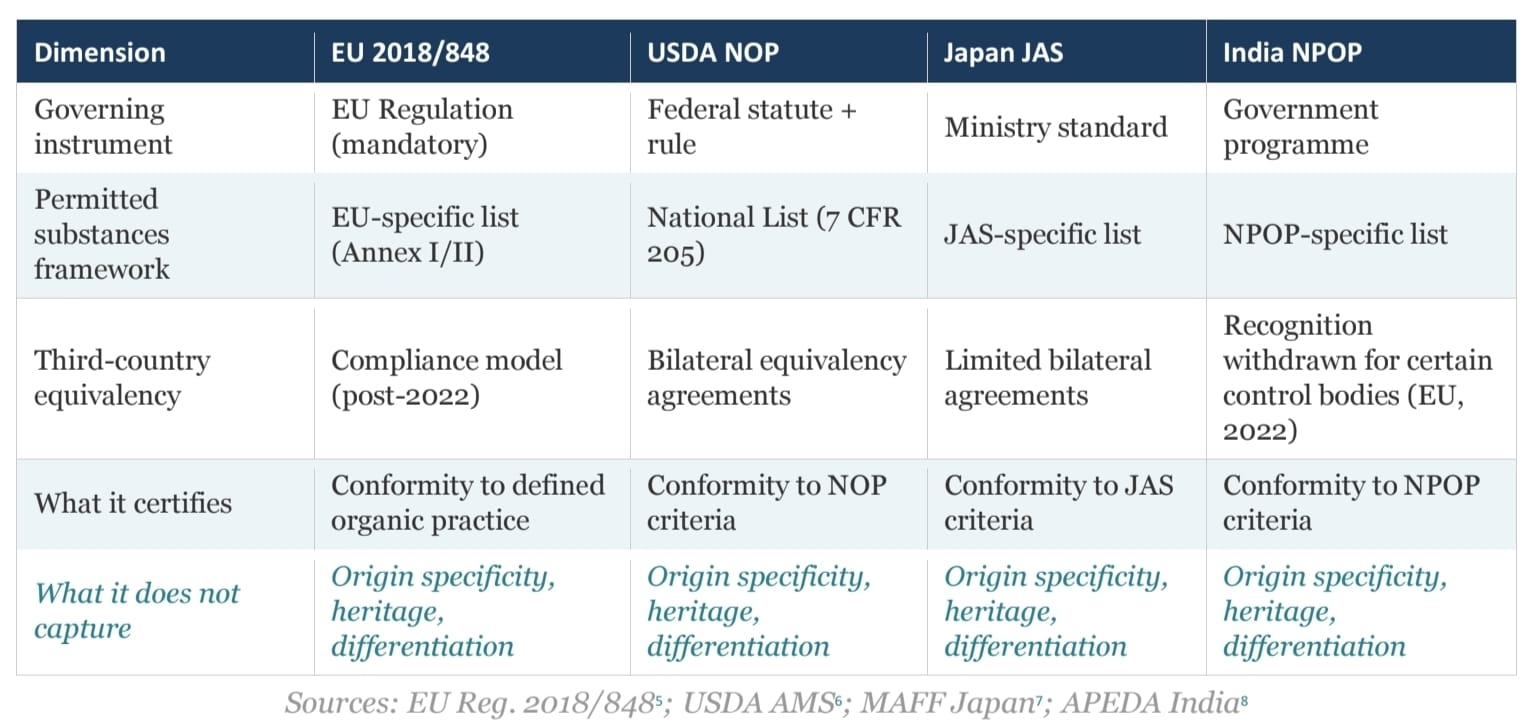
Sources: EU Reg. 2018/848⁵; USDA AMS⁶; MAFF Japan⁷; APEDA India⁸
The final row is identical across all four systems — by design. Capturing origin specificity, heritage, or qualitative differentiation is outside the scope of each, not an oversight within it.
The Codex Alimentarius Guidelines (CAC/GL 32-1999) function as a non-binding reference floor.⁹ Their purpose is to reduce the risk that national organic standards diverge so far as to become mutually incomprehensible — not to establish cross-system intelligibility. The guidelines have not prevented the permitted substances divergence noted above, nor the equivalency fragility illustrated by the India–EU case.
The Permitted Substances Gap
The USDA National List and the EU's permitted substances annexes differ in specific inputs that are allowed in one system and restricted or prohibited in the other. This divergence is not an administrative anomaly — it reflects different scientific advisory processes, precautionary standards, and agricultural contexts. Its practical consequence for an exporter holding both NOP and EU organic certification is that practices permissible for one destination may need to be adjusted or separately documented for the other. The certification marks held are not mutually legible.
Asymmetric Burden on Smaller Producers
Research published through the Research Institute of Organic Agriculture (FiBL) documents a consistent pattern: certification costs as a proportion of farm revenue tend to be higher for smaller operations than for larger ones, a relationship shaped by the fixed costs of inspection, recordkeeping, and renewal that do not scale proportionally with farm size.³ Group certification schemes — intended to reduce per-producer cost by distributing inspection load across cooperatives or associations — introduce their own coordination requirements that can exclude the most geographically dispersed or administratively under-resourced producers.
The structural result is that the certified sector does not uniformly represent the population of producers whose practices substantively meet organic standards. Available evidence suggests that some producers whose methods align with the substantive intent of a scheme standard remain outside the certified category due to cost and procedural barriers — a pattern documented in both emerging and established export markets.¹⁰,¹³ For trade agencies engaged in export capacity building, this represents a structural gap between production capability and documented market access.
Threshold Architecture and Certification Scope
Where certification operates as a market-access requirement — a gate that must be cleared to enter a premium segment or trade corridor — producers direct compliance effort toward auditable minimum criteria. This is a rational response to the incentive structure of threshold architecture, not a pathological one. It is not a critique of producers or certifiers. It is an observation that the architecture of a threshold instrument shapes the documentation activity it elicits: activity oriented toward meeting the threshold, rather than toward capturing the fuller range of producer knowledge that exists beyond it.
A producer's knowledge of the terroir, seasonal variation, traditional methods, or micro-practice distinctions that define their product does not enter the certification record. That producer knowledge exists at the farm level. The certification architecture has no mechanism to transmit it.
4. Implications
For trade agencies and export promotion bodies, the structural dynamics described in this paper carry three operational consequences.
First: certification-to-certification translation remains an active coordination cost. Where a producing country's certification system lacks equivalency recognition in a target destination market — as India–EU demonstrates — exporters face parallel certification requirements for the same product. This reflects the architecture of independently developed national systems, and equivalency arrangements, where they exist, require active negotiation and maintenance to remain in force.¹² Trade agencies involved in market-access work have an ongoing need to map the certification landscape of destination markets, not only the tariff and regulatory dimensions of trade.
Second: the gap between threshold and differentiation constrains value communication. Where certification is the primary informational signal available to buyers, competitive differentiation among certified producers is structurally compressed. Producer-level characteristics — heritage, origin specificity, practice distinction — that might otherwise be legible to buyers and support premium positioning remain undocumented in any form that travels with the product. Export promotion bodies whose mandate includes value capture alongside market access confront a structural obstacle that sits outside the scope of certification policy: the absence of an information layer that operates beyond the threshold.
Third: documentation redundancy creates a compounding burden on smaller exporters. As the number of destination markets a producer serves increases, the cost of maintaining destination-specific certification stacks rises — not because practices change, but because each system's proof architecture is independent. This asymmetry is not uniform: larger exporters can absorb multi-system certification cost as an operational line item. For smaller producers and cooperatives in emerging export markets, the same cost is proportionally larger and may determine whether multi-market access is viable at all.
These consequences arise from the structural design of certification systems, not from their implementation quality. They are not addressable through improvements to individual certification programmes. They require attention at a different informational layer.
5. Toward Structural Alternatives
The structural gap identified in this paper — between what certification establishes and what buyers, trade institutions, and regulators increasingly need — is not closed by improving certification systems. Certification performs its defined function well. The gap exists at the layer of producer-level information that sits above and beyond the threshold event: origin specificity, practice narrative, heritage context, seasonal variation, and the qualitative characteristics that distinguish producers within the same certified category.
Any information architecture capable of operating at that layer would need to satisfy several structural requirements, which can be stated independently of any specific system or model:
It would need to capture producer-level information beyond the binary threshold event — including origin specificity, production method detail, contextual variation, and heritage practice — in a form that is attributable to the producer, structured for comparability, and persistent over time.
It would need to be legible across jurisdictions without creating new equivalency dependencies — enabling cross-border comparability through shared information structure rather than through the negotiation of bilateral recognition arrangements.
It would need to operate on a principle of attribution: distinguishing clearly between what the producer has declared and what constitutes general contextual background, so that institutional readers can calibrate their interpretation accordingly. This is a different function from verification — it does not establish truth, it establishes source.
It would need to function as a complement to existing certification systems, not a replacement for them. Baseline assurance — the function certification performs — remains genuinely valuable. The informational need described here operates above the threshold, not instead of it.
It would need to be accessible to producers of varying scale — including those for whom multi-certification maintenance is already a disproportionate burden. An architecture that replicates those barriers would reach the same segment of the producer population that existing systems already serve.
This paper does not propose a specific model. It outlines structural requirements that any viable alternative would need to satisfy.
References
-
Hemmerling, S., Hamm, U., & Spiller, A. (2015). Consumption behaviour regarding organic food from a marketing perspective — a literature review. Organic Agriculture, 5(4), 277–313.
-
Codex Alimentarius Commission. (1999, rev. 2013). Guidelines for the Production, Processing, Labelling and Marketing of Organically Produced Foods. CAC/GL 32-1999. FAO/WHO, Rome/Geneva.
-
Willer, H., Trávníček, J., Meier, C., & Schlatter, B. (Eds.). (2024). The World of Organic Agriculture: Statistics and Emerging Trends 2024. FiBL & IFOAM – Organics International, Frick and Bonn.
-
European Commission. (2022). Commission Implementing Regulation (EU) 2022/2049 of 24 October 2022 amending Implementing Regulation (EU) 2021/2325 as regards the withdrawal of the recognition of certain third countries and control authorities and control bodies for the purpose of importing organic products into the Union. Official Journal of the European Union, L 275, 64–77.
-
European Parliament and Council. (2018). Regulation (EU) 2018/848 on organic production and labelling of organic products. Official Journal of the European Union, L 150/1.
-
United States Department of Agriculture, Agricultural Marketing Service. (2000, as amended). National Organic Program: National List of Allowed and Prohibited Substances. 7 CFR § 205.600–205.606. Washington, D.C.
-
Ministry of Agriculture, Forestry and Fisheries, Japan. (2022). Japan Agricultural Standard for Organic Plants (JAS 0059:2022, as revised). Tokyo: MAFF. Note: subsequent revisions to the JAS organic standard family were issued in 2024–2025; this citation refers to the 2022 version of the standard as the basis for the cross-system comparison in Section 3.
-
Agricultural and Processed Food Products Export Development Authority (APEDA). National Programme for Organic Production (NPOP): Standards and Accreditation. New Delhi: Ministry of Commerce and Industry, Government of India. [Current version]
-
Codex Alimentarius Commission. (1999, rev. 2013). Op. cit.
-
International Trade Centre (ITC). (2019). The State of Sustainable Markets: Statistics and Emerging Trends 2019. ITC, Geneva.
-
European Commission. (2021). EU Action Plan for the Development of Organic Production. COM(2021) 141 final. Brussels.
-
IFOAM – Organics International. (2023). IFOAM Equivalence Service: Overview and Methodology. Bonn.
-
FAO. (2021). Organic Agriculture: Meeting Global Demand While Ensuring Food Safety and Environmental Sustainability. Food and Agriculture Organization, Rome.
Version 1.0 — March 2026
This paper presents structural analysis of food information systems for institutional and research purposes. It does not constitute legal advice, regulatory guidance, certification assessment, or policy recommendation. The observations presented describe informational architecture, not evaluations of specific certification bodies, national programmes, or regulatory authorities.
Published by Altibbe Inc. | altibbe.com/research
Download the official PDF: SGPIS-GQ-01.pdf